Understanding chemical bonding is essential in chemistry as it helps to explain the process by which atoms come together to create molecules and compounds. It is the fundamental force responsible for holding atoms together, creating everything from the smallest molecules of air to the complex structures of living organisms.
This article explores chemical bonding in greater depth, covering its types, properties, and crucial role in the physical and biological world. You can learn step-by-step guides in the USA from our online trusted website Nurse Preceptors.
What is Chemical Bonding?
Chemical bonding is the process by which atoms join together to form compounds and molecules. It involves the interaction of the outermost electrons of atoms—known as valence electrons. Atoms bond to achieve a more stable electron configuration, often resembling the electron configuration of chemically inert noble gases.
Atoms typically bond because the resulting molecule or compound is more stable than the individual atoms. The stability is often due to the atom achieving a full outer shell of electrons, a state referred to as the octet rule in many cases. While this rule is common, it is not universal, especially for elements beyond the second row of the periodic table, where expanded valence shells and other bonding behaviors occur.
Chemical bonds can be divided into two major categories: Intramolecular bonding and Intermolecular bonding. These types of bonding contribute to the structure, properties, and reactivity of chemical compounds.
What are the Types of Chemical Bonding?
- There are 2 main types of chemical bonding:
- Intramolecular Bonding
- Intermolecular Bonding
1. Intramolecular Bonding
The forces that keep atoms bonded within a molecule are known as intramolecular bonds. These bonds form the backbone of the molecule and significantly determine its shape, stability, and reactivity.
- Intramolecular bonds are further divided into three parts:
- A. Ionic Bonding
- B. Covalent Bonding
- C. Metallic Bonding
A. Ionic Bonding
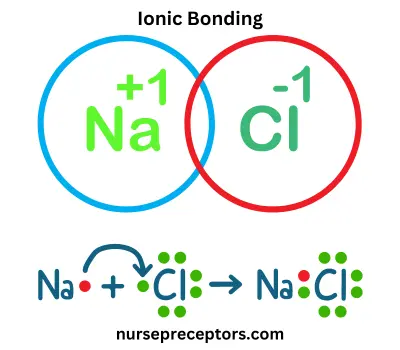
Ionic bonding occurs when electrons are transferred from one atom to another, resulting in the formation of ions (atoms with an electrical charge). This typically happens between metals and non-metals. When an atom loses electrons, it transforms into a positively charged ion known as a cation. On the other hand, an atom that gains electrons becomes a negatively charged ion, which is called an anion. The strong ionic bond is formed due to the electrostatic attraction between these oppositely charged ions.
Example: Sodium chloride (NaCl) is a classic ionic compound. Sodium (Na) donates an electron to chlorine (Cl), forming Na⁺ and Cl⁻ ions. These ions are subsequently bound by robust electrostatic forces.
- Ionic compounds generally exhibit:
- High melting and boiling points: Due to strong electrostatic attraction, a lot of energy is required to break these bonds.
- Hardness and brittleness: Ionic compounds form rigid crystalline lattices but can shatter when a force causes like-charged ions to repel.
- Electrical conductivity in molten or dissolved states: In a solid state, ionic compounds do not conduct electricity because the ions are fixed in place. When the substance is melted or dissolved in water, the ions gain mobility, which allows for the conduction of electricity.
B. Covalent Bonding
Covalent bonding is a fundamental type of chemical bonding characterized by the exchange of electron pairs between atoms. This mutual sharing of electrons facilitates the attainment of a complete outer electron shell, promoting greater stability for the atoms involved.
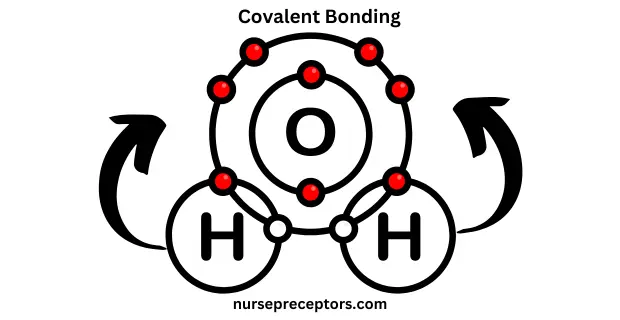
In covalent bonding, the shared electrons are typically contributed by both atoms, and the resulting bond can be single, double, or triple depending on the number of electron pairs shared. This type of bonding is commonly found in molecules and is responsible for the formation of many different compounds. This bond typically forms between non-metal atoms and results in the formation of molecules.
Example: Water (H₂O) is formed by covalent bonds. When oxygen bonds with two hydrogen atoms, it forms a stable molecule by sharing electrons. The sharing of electrons is unequal due to oxygen’s higher electronegativity, resulting in polar covalent bonds.
- Covalent bonds can be classified as:
- Non-polar covalent bonds: The equal sharing of electrons occurs between atoms. This usually happens between atoms of the same element, such as in diatomic molecules like O₂ or N₂.
- Polar covalent bonds: Electrons are shared unequally, creating a dipole moment where one side of the molecule is slightly negative and the other side is somewhat positive. Water (H₂O) is a prime example of a molecule with polar covalent bonds.
- Covalent compounds often:
- Have lower melting and boiling points than ionic compounds: This is because the forces holding the molecules together (intermolecular forces) are generally weaker than the electrostatic forces in ionic compounds.
- Exist as gases, liquids, or soft solids at room temperature: Unlike the rigid crystalline structures of ionic compounds, covalent compounds can take various forms.
- Do not conduct electricity: Since covalent molecules do not consist of charged particles, they do not conduct electricity in solid, liquid, or dissolved states.
C. Metallic Bonding
Metallic bonding occurs in metals, where valence electrons are not bound to any specific atom. Instead, these electrons form a “sea of electrons” that flow freely around the metal atoms. This delocalized electron cloud allows metals to exhibit several characteristic properties.
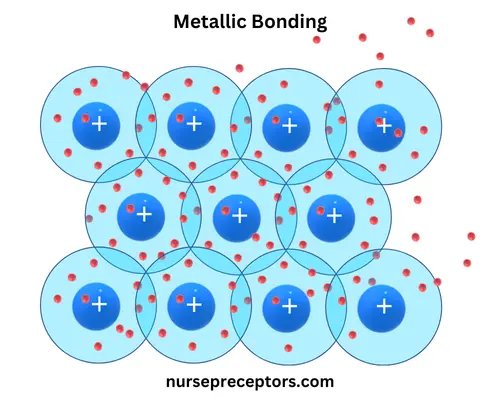
Example: Copper (Cu) is held together by metallic bonds, which allow electrons to move freely, contributing to its conductivity and malleability.
- Metallic bonding gives rise to:
- High electrical and thermal conductivity: The free movement of electrons allows metals to conduct electricity and heat efficiently.
- Malleability and ductility: Metals can be hammered into thin sheets (malleability) or drawn into wires (ductility) because the metal atoms can slide past each other without breaking the bond.
- Luster: The delocalized electrons reflect light, giving metals their shiny appearance.
The role of the Pakistan Nursing Council (PNC) in shaping nursing education.
2. Intermolecular Bonding
Intermolecular bonds are forces of attraction between molecules rather than within them. Although it’s weaker than intramolecular bonds, intermolecular forces are vital for determining many physical properties of substances, such as boiling points, melting points, and solubility. The main types of intermolecular forces include:
A. Van der Waals Forces
Van der Waals forces, also known as London dispersion forces, are weak intermolecular forces that arise from temporary dipoles induced in atoms or molecules. These forces are present in all molecules, but they are especially significant in non-polar molecules.
Example: Noble gases like argon (Ar) and molecules like methane (CH₄) exhibit Van der Waals forces.
- Characteristics of Van der Waals forces include:
- Weak and short-range: These forces are the weakest form of intermolecular attraction and act only over short distances.
- Influence on physical states: Though weak, Van der Waals forces are responsible for the condensation of non-polar substances into liquids and solids at low temperatures.
What is the fundamental concept behind the definition of nursing?
B. Dipole-Dipole Interactions
Dipole-dipole interactions occur between molecules that have permanent dipoles, i.e., polar molecules. These molecules are attracted to each other due to the positive end of one molecule being attracted to the negative end of another.
Example: Hydrogen chloride (HCl) molecules are polar, with a partial positive charge on hydrogen and a partial negative charge on chlorine, leading to dipole-dipole interactions.
- These interactions lead to:
- Higher melting and boiling points for polar substances compared to non-polar substances of similar molecular mass.
- Solubility in polar solvents, as polar molecules interact strongly with other polar molecules like water.
What are the Benefits and Limitations of Gordon’s Functional Health Patterns?
C. Hydrogen Bonding
Hydrogen bonding is a special and stronger type of dipole-dipole interaction that occurs when hydrogen is covalently bonded to a highly electronegative atom (nitrogen, oxygen, or fluorine).
The hydrogen atom carries a partial positive charge, which is attracted to the partial negative charge on a nearby electronegative atom in another molecule.
Example: Water (H₂O) molecules form hydrogen bonds between the hydrogen atom of one molecule and the oxygen atom of another, which gives water its unique properties.
- Hydrogen bonding is responsible for:
- High boiling and melting points of water: Despite its low molecular weight, water remains a liquid over a wide temperature range because of strong hydrogen bonds.
- Unusual properties of ice: In its solid state, water molecules form a crystalline structure that is less dense than liquid water, making ice float.
- Structural integrity of biological molecules: Hydrogen bonding is critical in maintaining the three-dimensional structure of proteins, DNA, and RNA.
What are the Properties of Chemical Bonds?
Chemical bonds, whether intramolecular or intermolecular, dictate many of the physical and chemical properties of substances. These properties include:
1. Bond Strength
It’s important to grasp the concept of bond strength since it reflects the energy required to break a bond. Intramolecular bonds such as ionic, covalent, and metallic bonds generally exhibit greater strength compared to intermolecular forces like Van der Waals forces, dipole-dipole interactions, and hydrogen bonds.
- Strong bonds, such as ionic and covalent bonds, give substances higher melting and boiling points.
- Weak bonds, such as Van der Waals forces, are easily disrupted, leading to lower melting and boiling points.
2. Bond Length
The bond length is defined as the distance between the nuclei of two atoms that are chemically bonded. It is determined by the type of bond and the size of the atoms involved. Generally, shorter bonds are stronger as they hold the atoms more tightly together.
- Covalent bonds: Generally, have shorter bond lengths, especially in molecules with double or triple bonds.
- Ionic bonds: Tend to have longer bond lengths due to the larger radii of the ions involved.
3. Bond Polarity
The concept of bond polarity revolves around the arrangement of electron density within a bond. In cases where two atoms with varying electronegativities come together to form a bond, there is an uneven sharing of electrons, leading to the formation of a polar bond. It’s worth noting that the larger the difference in electronegativity, the greater the polarity of the bond.
- Non-polar bonds: Occur between atoms of similar electronegativity, such as in O₂.
- Polar bonds: Result in a separation of charge and occur in molecules like H₂O, where oxygen has a higher electronegativity than hydrogen.
4. Melting and Boiling Points
The melting and boiling points of a substance are directly influenced by the strength of the bonds within it. For instance, substances with strong bonds, like ionic and covalent solids, need a higher amount of energy to change from a solid to a liquid or from a liquid to a gas state.
- Ionic compounds: Ionic compounds generally exhibit high melting and boiling points as a result of the robust electrostatic forces between ions.
- Covalent compounds: Usually have lower melting and boiling points compared to ionic compounds, except when strong intermolecular forces (such as hydrogen bonding) are present.
5. Electrical Conductivity
Electrical conductivity depends on the availability of free-moving charged particles.
- Ionic compounds: Conduct electricity in molten or aqueous states because the ions are free to move.
- Metals: Conduct electricity due to the presence of free-moving delocalized electrons.
- Covalent compounds: Do not typically conduct electricity, as they lack charged particles.
6. Solubility
Understanding solubility is crucial as it refers to a substance’s ability to dissolve in a specific solvent. Polar compounds tend to dissolve in polar solvents such as water, while non-polar compounds dissolve in non-polar solvents like hexane. This knowledge helps us predict how different substances will interact when mixed.
Importance of Chemical Bonding
The importance of chemical bonding cannot be overstated. It is fundamental to both the natural and synthetic worlds, playing a role in everything from the simplest compounds to complex biological structures.
1. Creation of Matter
Chemical bonding is essential for the formation of compounds and molecules, which in turn make up all forms of matter. Whether it’s the salt in your food or the DNA in your cells, bonding holds these structures together.
2. Determination of Physical and Chemical Properties
The type of bonding in a substance determines many of its physical properties, such as hardness, melting and boiling points, and solubility. These properties are key to the application of materials in various industries.
3. Vital Biological Processes
In living organisms, chemical bonds play a crucial role in maintaining the structure and function of biomolecules.
- For example:
- DNA’s double helix is stabilized by hydrogen bonds between complementary base pairs.
- Protein folding depends on a combination of covalent and non-covalent bonds, including hydrogen bonds, disulfide bridges, and Van der Waals interactions.
- Enzyme-substrate interactions, essential for metabolic processes, often involve weak bonds that can form and break easily during reactions.
4. Material Science and Technology
The ability to manipulate bonding is central to the development of new materials with specific properties. Advances in nanotechnology, pharmaceuticals, and synthetic polymers rely on precise control over chemical bonding at the atomic and molecular levels.
Superconductors, materials that conduct electricity without resistance, depend on specific types of bonding.
Smart materials, such as shape-memory alloys and hydrogels, exploit chemical bonds that respond to changes in temperature, pressure, or other environmental conditions.
5. Environmental and Chemical Reactions
Chemical bonds also govern the behavior of substances in chemical reactions, influencing the energy required for reactions to occur and the stability of the resulting products. For instance, combustion, photosynthesis, and respiration all involve the making and breaking of chemical bonds, releasing or absorbing energy in the process.
Conclusion
Chemical bonding is the invisible force that drives the formation of molecules, the behavior of materials, and the intricate processes that sustain life. From the strength of metals to the flexibility of proteins, every substance’s properties can be traced back to the type of bonds holding its atoms together.
By understanding chemical bonding, we gain insights into the workings of the universe on an atomic level, allowing us to predict, manipulate, and create materials and systems for countless applications in science, industry, and medicine.
Chemical bonding is the key to the molecular world, shaping everything from the air we breathe to the technology we use, making it a subject of perpetual importance and exploration in both science and everyday life.
References:
- Chemical bonding – The law of conservation of mass. (n.d.). Encyclopedia Britannica. https://www.britannica.com/science/chemical-bonding/The-law-of-conservation-of-mass
- Chemical Bonding – Types of Chemical Bonds, Bond Characteristics, Enthalpy. (n.d.). BYJUS. https://byjus.com/jee/chemical-bonding/
- Wikipedia Contributors. (2019, November 15). Chemical bond. Wikipedia; Wikimedia Foundation. https://en.wikipedia.org/wiki/Chemical_bond
- Arif Malik, M. (2022, April 5). 3.9: Intramolecular forces and intermolecular forces. Chemistry LibreTexts. https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Introduction_to_General_Chemistry_(Malik)/03%3A_Compounds/3.09%3A_Intramolecular_forces_and_intermolecular_forces
- Intramolecular Forces. (n.d.). Www.simply.science. https://www.simply.science/images/content/chemistry/states_of_matter/force_of_attraction/conceptmap/intra_molecular_forces.html
- Intramolecular bonding – Structure and bonding – Higher Chemistry Revision. (n.d.). BBC Bitesize. https://www.bbc.co.uk/bitesize/guides/zt9887h/revision/1
- Ionic Bond (Electrovalent Bond) – Definition, Properties, Electronegativity & Examples with Videos. (n.d.). BYJUS. https://byjus.com/chemistry/ionic-bond-or-electrovalent-bond/
- Science Direct. (2010). Covalent Bond – an Overview | ScienceDirect Topics. Sciencedirect.com. https://www.sciencedirect.com/topics/chemistry/covalent-bond
- Clark, J. (2012). metallic bonding. Chemguide.co.uk. https://www.chemguide.co.uk/atoms/bonding/metallic.html
- to, C. (2012, April 15). Residual attractive or repulsive forces between molecules or atomic groups that do not arise from a covalent bond or ionic bond. Wikipedia.org; Wikimedia Foundation, Inc. https://simple.wikipedia.org/wiki/Van_der_Waals_force
- Dipole-Dipole Forces. (2019). Purdue.edu. https://www.chem.purdue.edu/gchelp/liquids/dipdip.html
- Properties: Properties of Chemical Bonds. (n.d.). SparkNotes. https://www.sparknotes.com/chemistry/bonding/properties/section1/
- Gudzuadze, M. (2022, June 22). 10 Reasons Why Chemical Bonds Are Important. The Important Site. https://theimportantsite.com/reasons-why-chemical-bonds-are-important/
- Gudzuadze, M. (2022, June 22). 10 Reasons Why Chemical Bonds Are Important. The Important Site. https://theimportantsite.com/reasons-why-chemical-bonds-are-important/
- Gupta, P. (2020). Chemical Bonding: Importance and Conceptions. https://www.rroij.com/open-access/chemical-bonding-importance-and-conceptions.pdf
- Constable, E. C., & Housecroft, C. E. (2020). Chemical Bonding: The Journey from Miniature Hooks to Density Functional Theory. Molecules, 25(11), 2623. https://doi.org/10.3390/molecules25112623
- What is the importance of chemical bonding in chemistry and in our life – All the compounds ( both – Studocu. (2021). Studocu; Studocu. https://www.studocu.com/en-us/document/harvard-university/chemistry/what-is-the-importance-of-chemical-bonding-in-chemistry-and-in-our-life/27542204
- Types and significance of chemical bonds. (n.d.). https://stjohnscollege.edu.in/web/wp-content/uploads/2022/01/Types-and-significance-of-chemical-Bonds.pdf